| Availability: | |
|---|---|
| Quantity: | |











The Lumineer Semi-Compliant PTCA Balloon Catheter is a specialized interventional device designed for pre-dilatation and drug/stent delivery procedures. Equipped with a unique taper-tip design and upgraded TranSmooth™ II technology, this semi-compliant catheter balances flexibility and controlled expansion to meet the demands of complex vascular interventions. Its thin-wall balloon construction and high-visibility radiopaque marker bands deliver superior traceability and travelability, even when coated with drugs or crimped with stents. Unlike non-compliant alternatives, its semi-compliant nature adapts slightly to vessel contours while maintaining expansion accuracy, making it a preferred choice for cardiologists focused on efficient drug and stent delivery.
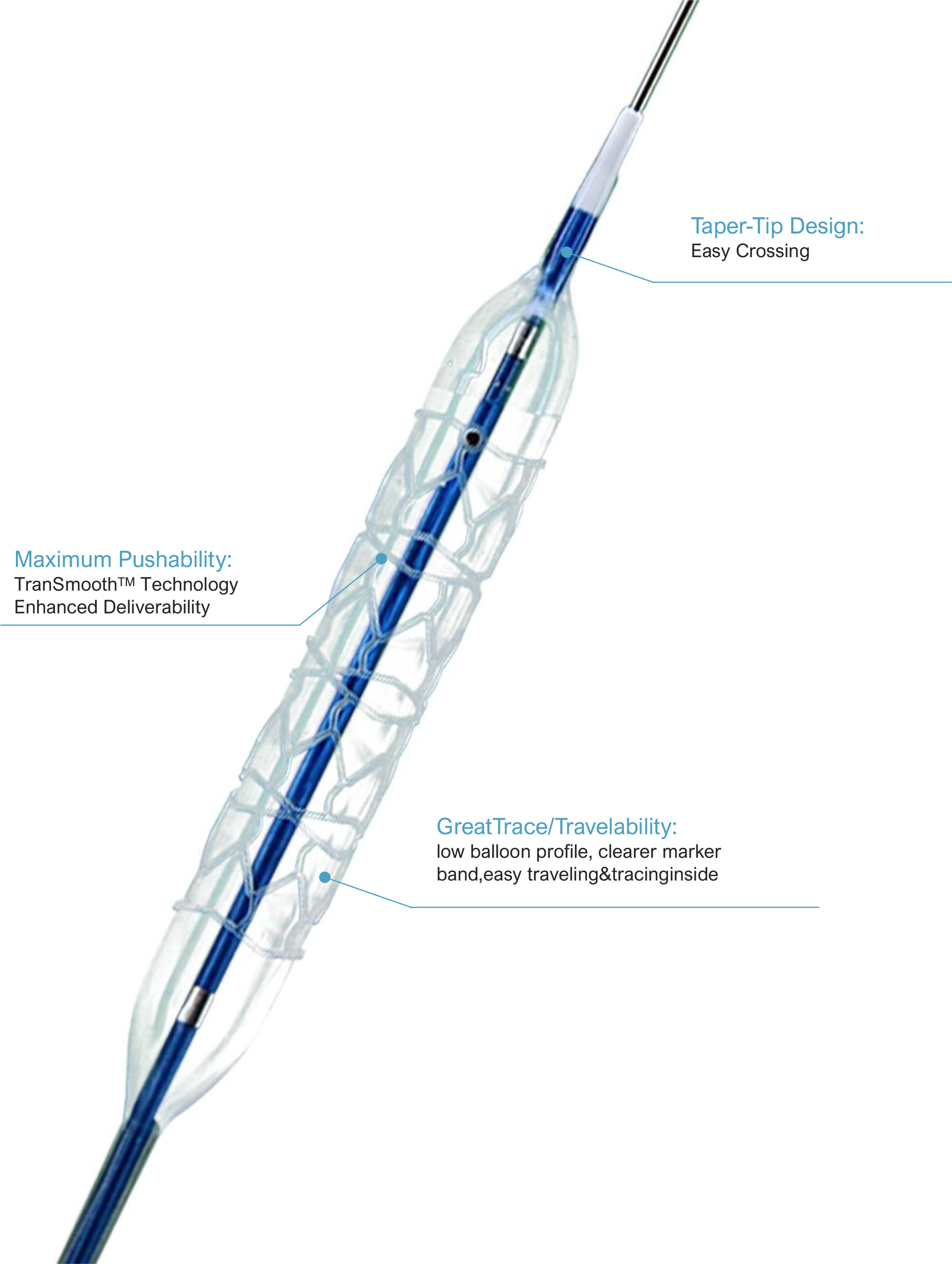
The taper-tip design features a gradual diameter transition, enabling easy navigation through narrow, tortuous vessels and calcified lesions. This reduces procedural time and minimizes trauma to vessel walls, improving patient safety.
Thin-wall balloon construction and high-density marker bands ensure excellent visibility under fluoroscopy, even when paired with drug coatings or stents. This allows cardiologists to track the catheter’s position with unparalleled accuracy.
The semi-compliant nature adapts gently to vessel contours while maintaining precise length and diameter control. This optimizes drug distribution for coated balloons and ensures uniform stent apposition, boosting treatment efficacy.
TranSmooth™ II technology enhances shaft flexibility and push force transmission, allowing seamless delivery through challenging vascular pathways such as coronary bifurcations.
Designed for pre-dilatation of vascular lesions to create optimal conditions for subsequent drug-eluting stent (DES) placement, ensuring the stent fits securely and functions effectively.
Ideal for delivering drug-coated balloons and stents to target lesion sites, its low-profile design and traceability ensure uniform drug contact with vessel walls, maximizing therapeutic effects.
Specialized for patients with tortuous or small-caliber vessels that require flexible, trackable interventional devices, addressing anatomical challenges that limit conventional catheters.
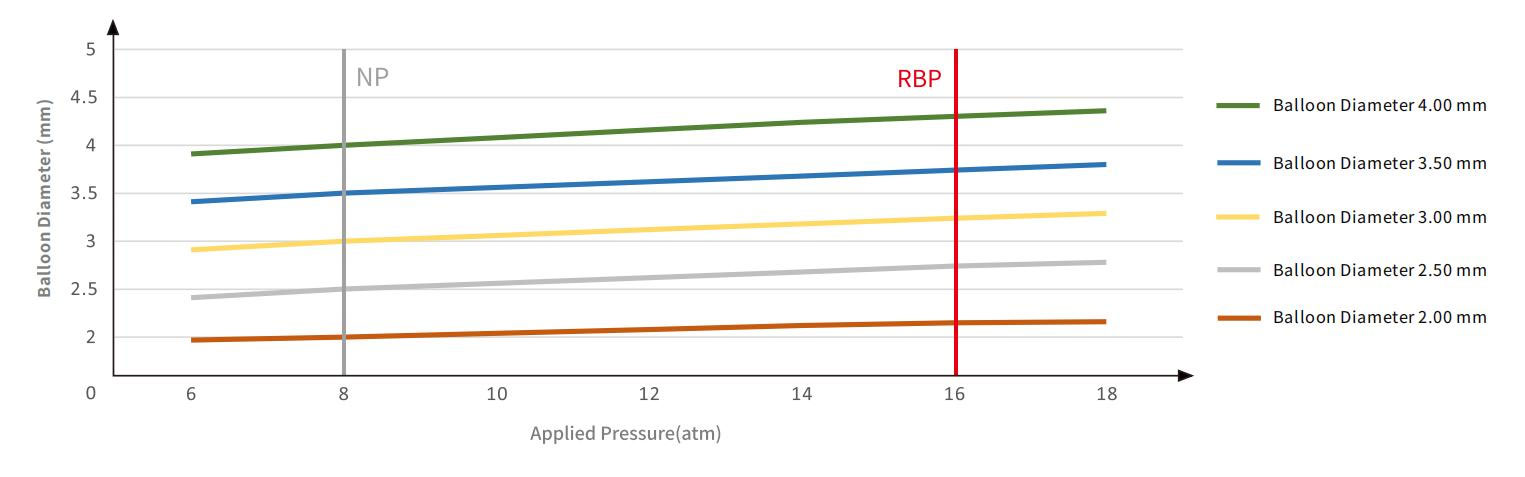
Lumineer Compliance chart
Semi-Compliant PTCA Balloon Catheter Designed Specifically for Pre-dilation and Drug / Stent Delivery
Technical Information
Lumineer Semi-Compliant PTCA Balloon Catheter | |
Platform | Rapid Exchange(Rx) |
Balloon Material | PA |
Marker Material | Platinum-Iridium |
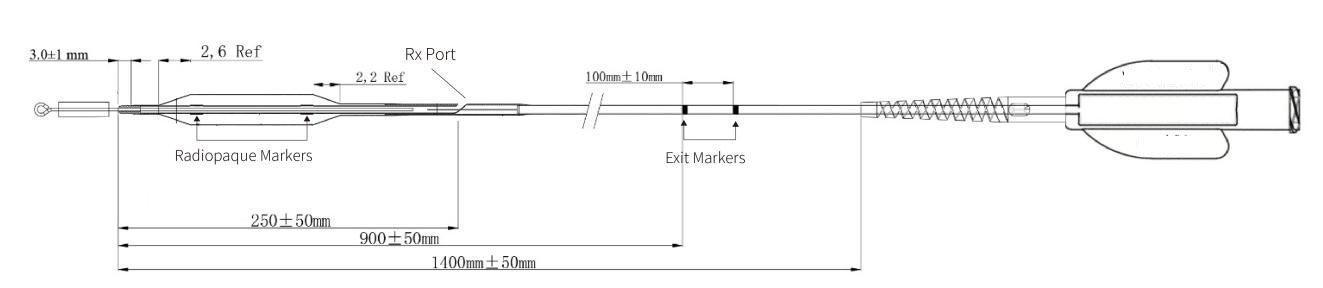
Tip Length | 3±1mm |
Distal Shaft Diameter | 0.89±0.05mm |
Proximal Shaft Diameter | 0.67±0.05mm |
Compliance | Semi-compliant |
Nominal Pressure(NP) | 8 atm |
Rated Burst Pressure(RBP) | 16 atm |
Catheter Shaft Length | 140±5cm |
Maximum Guide Wire Compatibility | 0.014inch |
Shelf Life | 24 months |
Tip Entry Profile | 0.018 inch |
Balloon Crossing Profle | 2.00-2.25:0.80 mm 2.50-2.75:0.85mm |
Minimum Guiding CatheterlD | 6F/5F |
Order Information
Lumineer Semi-Compliant PTCA Balloon Catheter Order Information | ||||||||
Balloon | Balloon Length(mm) | |||||||
10 | 14 | 18 | 22 | 26 | 30 | 34 | 38 | |
2.00 | SD2.00X10 | SD2.00X14 | SD2.00X18 | SD2.00X22 | SD2.00×26 | SD2.00X30 | SD2.00X34 | SD2.00X38 |
2.25 | SD2.25X10 | SD2.25X14 | SD2.25X18 | SD2.25X22 | SD2.25X26 | SD2.25X30 | SD2.25X34 | SD2.25X38 |
2.50 | SD2.50X10 | SD2.50X14 | SD2.50X18 | SD2.50X22 | SD2.50X26 | SD2.50X30 | SD2.50X34 | SD2.50X38 |
2.75 | SD2.75X10 | SD2.75X14 | SD2.75X18 | SD2.75X22 | SD2.75X26 | SD2.75X30 | SD2.75X34 | SD2.75X38 |
3.00 | SD3.00X10 | SD3.00X14 | SD3.00X18 | SD3.00X22 | SD3.00X26 | SD3.00X30 | SD3.00X34 | SD3.00X38 |
3.25 | SD3.25X10 | SD3.25X14 | SD3.25X18 | SD3.25X22 | SD3.25×26 | SD3.25X30 | SD3.25X34 | SD3.25X38 |
3.50 | SD3.50X10 | SD3.50X14 | SD3.50X18 | SD3.50X22 | SD3.50X26 | SD3.50X30 | SD3.50X34 | SD3.50X38 |
3.75 | SD3.75X10 | SD3.75X14 | SD3.75X18 | SD3.75X22 | SD3.75X26 | SD3.75X30 | SD3.75X34 | SD3.75X38 |
4.00 | SD4.00X10 | SD4.00X14 | SD4.00X18 | SD4.00X22 | SD4.00X26 | SD4.00X30 | SD4.00X34 | SD4.00X38 |
The unique tapered tip sets our catheter apart from standard straight-tip alternatives, enabling easier access to hard-to-reach lesions and reducing the risk of vessel injury during navigation.
Its low-profile design and compatibility with drug coatings and stents streamline procedural workflows, reducing preparation time and improving clinical efficiency for busy cardiology departments.
Every Lumineer catheter undergoes rigorous quality control testing, including pressure resistance and dimensional accuracy checks, ensuring consistent performance across all clinical applications.
With a 24-month shelf life and broad compatibility, our catheter offers a cost-efficient option for healthcare facilities without compromising on quality or clinical outcomes.
Yes, the Lumineer Semi-Compliant PTCA Balloon Catheter’s low-profile design and excellent traceability make it highly suitable for pairing with drug-coated balloons to ensure effective drug delivery.
It has a nominal pressure (NP) of 8 atm and a rated burst pressure (RBP) of 16 atm, balancing safety and controlled expansion for various interventional procedures.
It is fully compatible with 0.014 inch guide wires, the standard size for most coronary intervention procedures, ensuring seamless integration with existing clinical equipment.
Upgrade your drug and stent delivery procedures with the flexible, high-performance Lumineer Semi-Compliant PTCA Balloon Catheter. Reach out to our team now to learn more about product availability, pricing and clinical support. Please note that this device is only available for sale to or on the order of a licensed physician—always follow the product labeling instructions for safe and effective use.
