| Availability: | |
|---|---|
| Quantity: | |











The HyperForm Non-Compliant PTCA Balloon Catheter is a high-performance interventional device engineered specifically for pre-drug-eluting balloon (Pre-DEB) dilatation and post-stenting dilatation procedures. Featuring the proprietary TranSmooth™ technology, this non-compliant balloon catheter delivers exceptional pushability and crossability, making it ideal for treating complex vascular lesions with precision. Its robust design ensures resistance to high pressure and rupture, while the precision-controlled diameter and length expansion guarantee that vessels are dilated exactly as required. With a minimal intro-profile and compatibility with small guiding catheters, this catheter stands out as a reliable tool for interventional cardiologists seeking consistent and safe clinical outcomes.
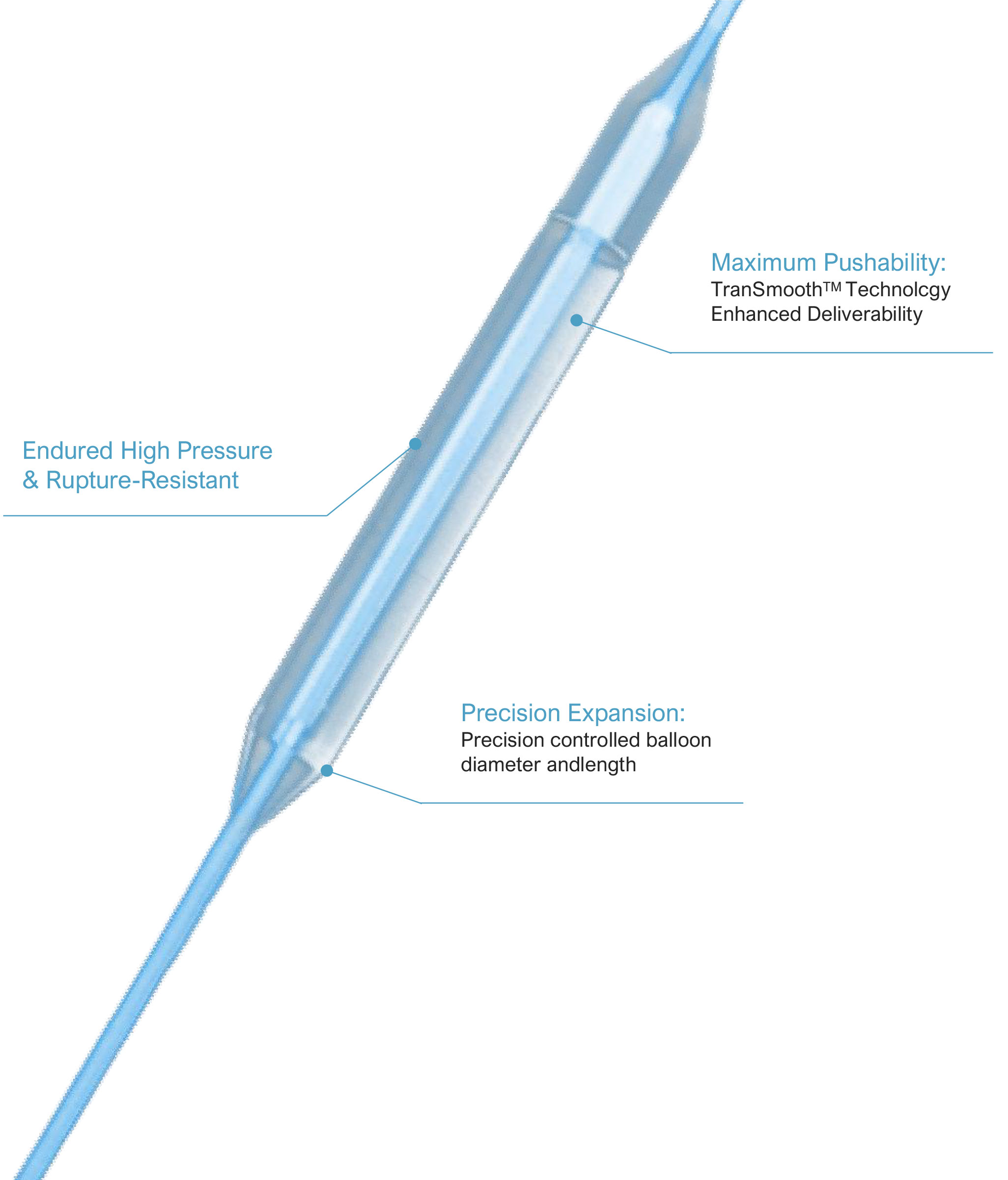
The catheter’s non-compliant nature ensures that balloon diameter and length remain stable under pressure, eliminating over-dilatation risks and ensuring vessels are treated as intended. This precision is critical for complex lesions where even minor deviations can affect clinical outcomes.
With a rated burst pressure of 18 atm, it endures high-pressure dilatation procedures without compromising structural integrity. This durability enhances procedural safety and gives cardiologists confidence when handling challenging cases.
The minimal tip entry profile and TranSmooth™ design maximize pushability, allowing easy navigation through small vessels and tortuous vascular pathways. This reduces procedural time and minimizes trauma to surrounding tissues.
Ideal for pre-dilatation before drug-eluting balloon (DEB) implantation, it prepares lesion sites to ensure optimal drug delivery and absorption, laying the groundwork for successful treatment.
Used for post-stenting dilatation to ensure full stent apposition against vessel walls, reducing restenosis risks and improving long-term vascular patency for patients.
Specialized for treating small vessel lesions that require precise, controlled dilatation without balloon over-expansion, addressing a common challenge in interventional cardiology.
The exclusive TranSmooth™ technology delivers unmatched pushability, ensuring smooth device delivery even in complex vascular anatomies that conventional catheters struggle to navigate.
Every catheter undergoes strict dimensional testing to guarantee consistency in diameter, length and pressure resistance, meeting global interventional device quality requirements and regulatory standards.
Compatible with 0.014 inch guide wires and 5F/6F guiding catheters, it offers flexibility for various clinical scenarios, adapting to different procedural needs without additional equipment investment.
A 24-month shelf life ensures inventory stability and reduces wastage for healthcare facilities, lowering operational costs while maintaining ready access to high-quality devices.
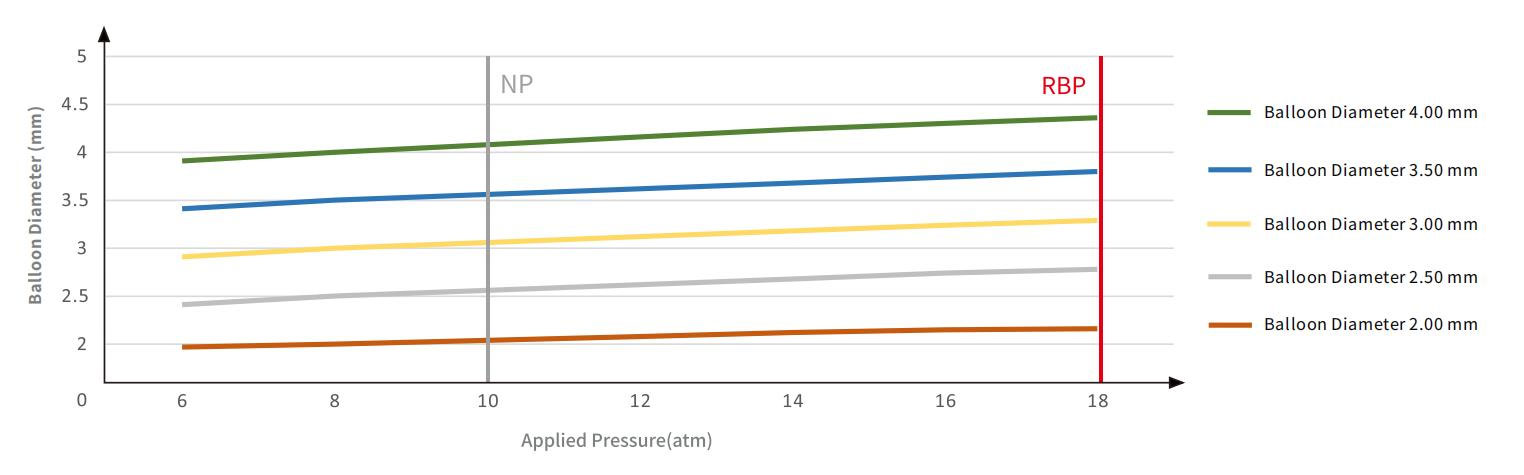
HyperForm Compliance Chart
Non-Compliant PTCA Balloon Cathcter Designed Specifically for Pre DEB or / and Post Stenting Dilatation
HyperForm Non-Compliant PTCA Balloon Catheter | ||||||||
Platform | Rapid Exchange(Rx) | |||||||
Balloon Material | PA | |||||||
Marker Material | Platinum-Iridium | |||||||
Tip Length | 3±1mm | |||||||
Distal Shaft Diameter | 0.89±0.05mm | |||||||
Proximal Shaft Diameter | 0.67±0.05 mm | |||||||
Compliance | Non-compliant | |||||||
Nominal Pressure(NP) | 10 atm | |||||||
Rated Burst Pressure(RBP) | 18 atm | |||||||
Catheter Shaft Length | 140±5cm | |||||||
Maximum Guide Wire Compatibility | 0.014 inch | |||||||
Shelf Life | 24 months | |||||||
Tip Entry Profile | 0.018inch | |||||||
Balloon Crossing Profile | 2.00-2.25:0.80mm 3.00-3.50:0.90mm | |||||||
Minimum Guiding CatheterID | 6F/5F | |||||||
HyperForm Non-Compliant PTCA Balloon Catheter Order Information | ||||||||
Balloon Diameter(mm) | Balloon Length(mm) | |||||||
10 | 14 | 18 | 22 | 26 | 30 | 34 | 38 | |
2.00 | NC2.00×10 | NC2.00×14 | NC2.00×18 | NC2.00×22 | NC2.00×26 | NC2.00×30 | NC2.00×34 | NC2.00×38 |
2.25 | NC2.25×10 | NC2.25×14 | NC2.25×18 | NC2.25×22 | NC2.25×26 | NC2.25×30 | NC2.25×34 | NC2.25×38 |
2.50 | NC2.50×10 | NC2.50×14 | NC2.50×18 | NC2.50×22 | NC2.50×26 | NC2.50×30 | NC2.50×34 | NC2.50×38 |
2.75 | NC2.75×10 | NC2.75×14 | NC2.75×18 | NC2.75×22 | NC2.75×26 | NC2.75×30 | NC2.75×34 | NC2.75×38 |
3.00 | NC3.00×10 | NC3.00×14 | NC3.00×18 | NC3.00×22 | NC3.00×26 | NC3.00×30 | NC3.00×34 | NC3.00×38 |
3.25 | NC3.25×10 | NC3.25×14 | NC3.25×18 | NC3.25×22 | NC3.25×26 | NC3.25×30 | NC3.25×34 | NC3.25×38 |
3.50 | NC3.50×10 | NC3.50×14 | NC3.50×18 | NC3.50×22 | NC3.50×26 | NC3.50×30 | NC3.50×34 | NC3.50×38 |
3.75 | NC3.75×10 | NC3.75×14 | NC3.75×18 | NC3.75×22 | NC3.75×26 | NC3.75×30 | NC3.75×34 | NC3.75×38 |
4.00 | NC4.00×10 | NC4.00×14 | NC4.00×18 | NC4.00×22 | NC4.00×26 | NC4.00×30 | NC4.00×34 | NC4.00×38 |
The HyperForm Non-Compliant PTCA Balloon Catheter has a rated burst pressure (RBP) of 18 atm, ensuring safe use under high-pressure dilatation conditions.
Yes, its minimal crossing profile (0.80mm–0.90mm) and non-compliant design make it ideal for small vessel lesions that demand precise dilatation without over-expansion.
The catheter has a shelf life of 24 months, providing ample time for storage and clinical use, which is beneficial for inventory planning.
Ready to elevate your vascular intervention procedures with a reliable, high-precision non-compliant PTCA balloon catheter? Contact our sales team today to inquire about pricing, availability and customization options. Note that this device is restricted to sale by or on the order of a physician—ensure proper training and adherence to product labeling instructions for optimal clinical outcomes.
