| Availability: | |
|---|---|
| Quantity: | |











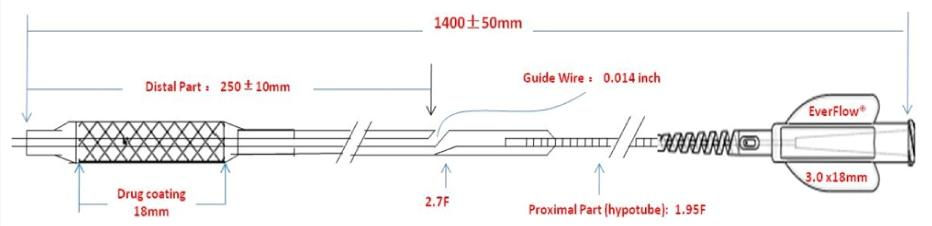
The Paclitaxel-Coated PTCA Balloon Catheter (brand name: NovaPac) is an innovative polymer-free drug delivery device designed for coronary artery interventions. It combines a drug-specific Lumineer balloon catheter with a 100% uniformly coated paclitaxel layer, delivering precision drug loading and controlled release without residual polymer. This system ensures minimal drug loss during delivery and achieves sustained arterial drug retention for up to 30 days post-balloon expansion, maintaining therapeutic paclitaxel concentrations within the safe and effective range (0.0085–0.85μg/mL). Preclinical studies in porcine coronary arteries demonstrate non-inferior performance compared to leading competitors, with no significant differences in lumen area (LA) and minimal lumen diameter (MLD) at 90 days follow-up. The semi-compliant balloon design and 2.7F distal profile enhance crossability, making it suitable for a wide range of coronary lesions.
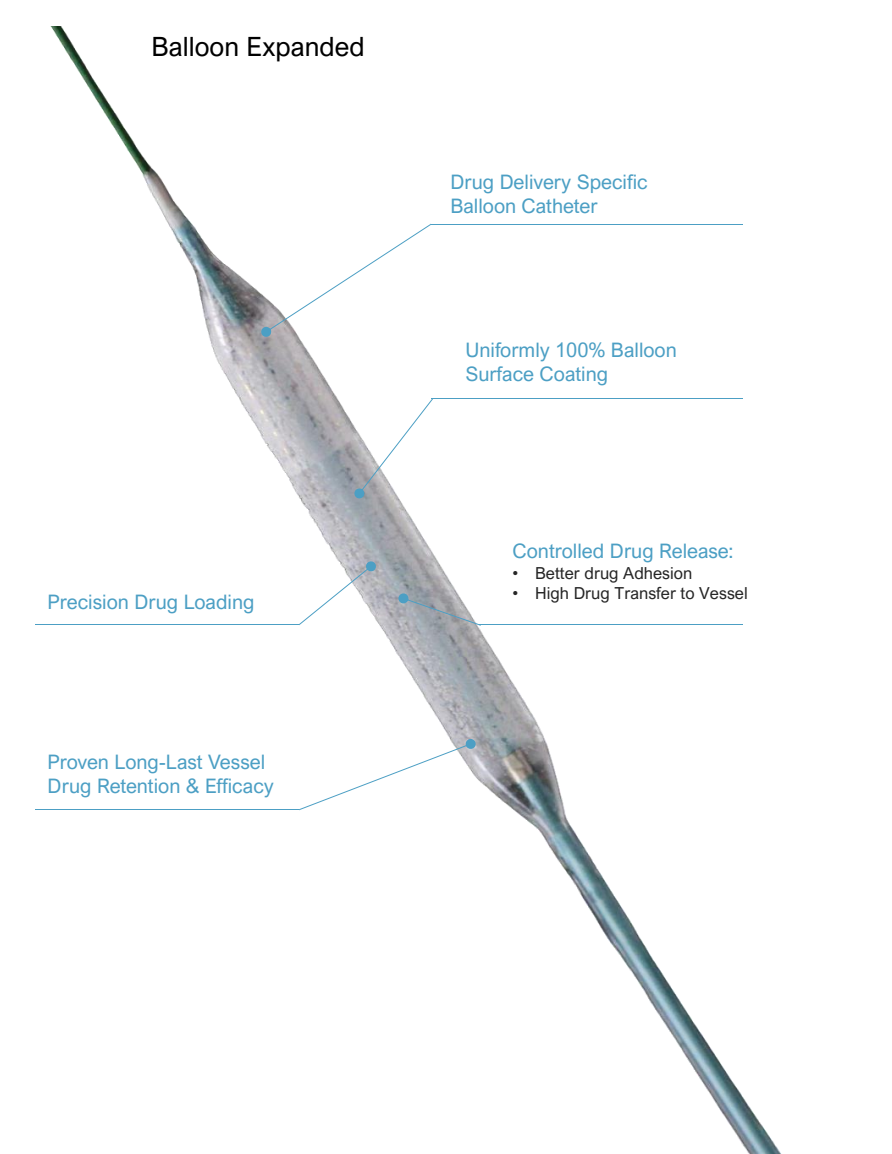
The proprietary coating technology ensures full balloon surface coverage with paclitaxel, eliminating polymer-related vascular irritation. This design enables precise drug loading and controlled release, maximizing drug transfer efficiency to the target vessel. The absence of residual polymer reduces long-term inflammatory responses, supporting healthier vascular recovery.
Effective paclitaxel concentrations remain in the treated vessel for up to 30 days post-expansion, providing sustained anti-restenotic effects. Pharmacokinetic studies confirm no detectable drug in core organs beyond 7 days, ensuring systemic safety while maintaining therapeutic efficacy at the lesion site. This balance of retention and safety enhances procedural outcomes.
The Lumineer balloon catheter features a semi-compliant design and low-profile distal shaft, enhancing navigation through narrow or tortuous coronary segments. Precision balloon expansion ensures uniform drug distribution across the lesion site, optimizing treatment effectiveness even in challenging anatomical conditions.
The Paclitaxel-Coated PTCA Balloon Catheter is ideal for primary stenosis, including small vessel disease (SVD) that requires targeted drug delivery without stent implantation. It delivers paclitaxel directly to the lesion, reducing restenosis risks in patients with de novo coronary artery lesions.
It effectively addresses post-stenting residual narrowing to optimize vessel patency. The catheter’s targeted drug delivery capability targets residual lesions, supplementing stent therapy and improving long-term vascular outcomes.
The catheter is used to prepare lesion sites before stent placement or optimize stent expansion after implantation. Its semi-compliant design and uniform coating ensure consistent performance in both pre and post-dilatation procedures, supporting seamless intervention workflows.
It provides rapid, localized drug delivery to reduce acute restenosis risks in critical occlusion scenarios. The quick-acting paclitaxel coating helps stabilize the vessel and prevent further occlusion, supporting urgent coronary intervention needs.
Evidence-Driven Device Safety and Efficacy
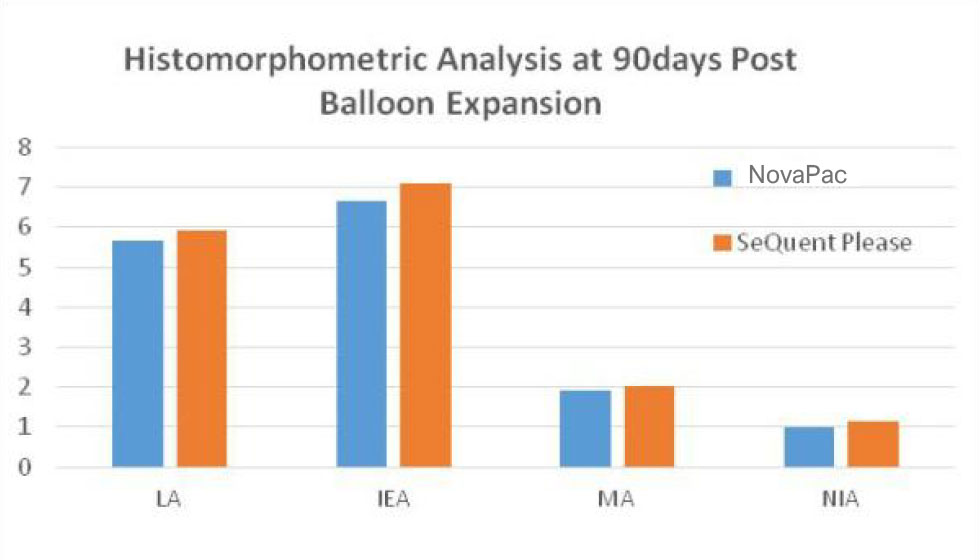
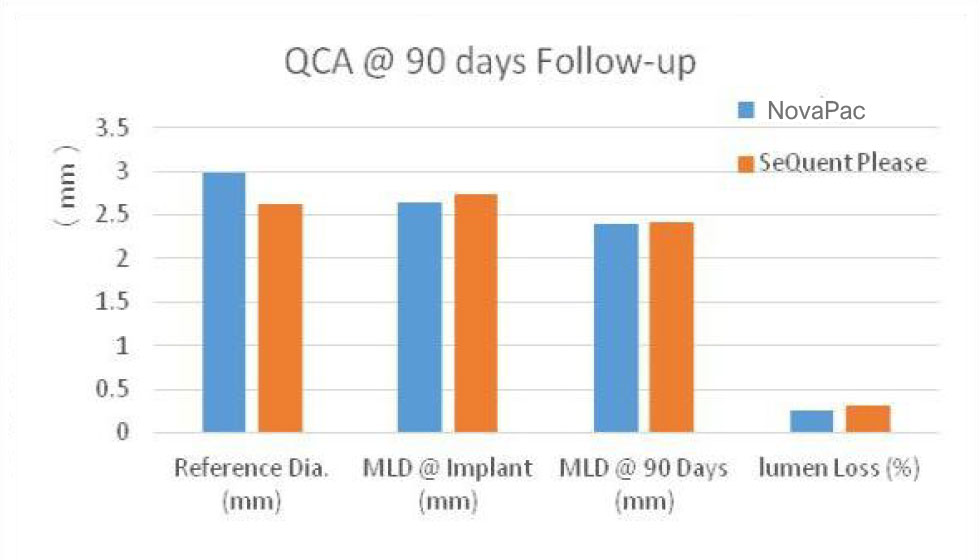
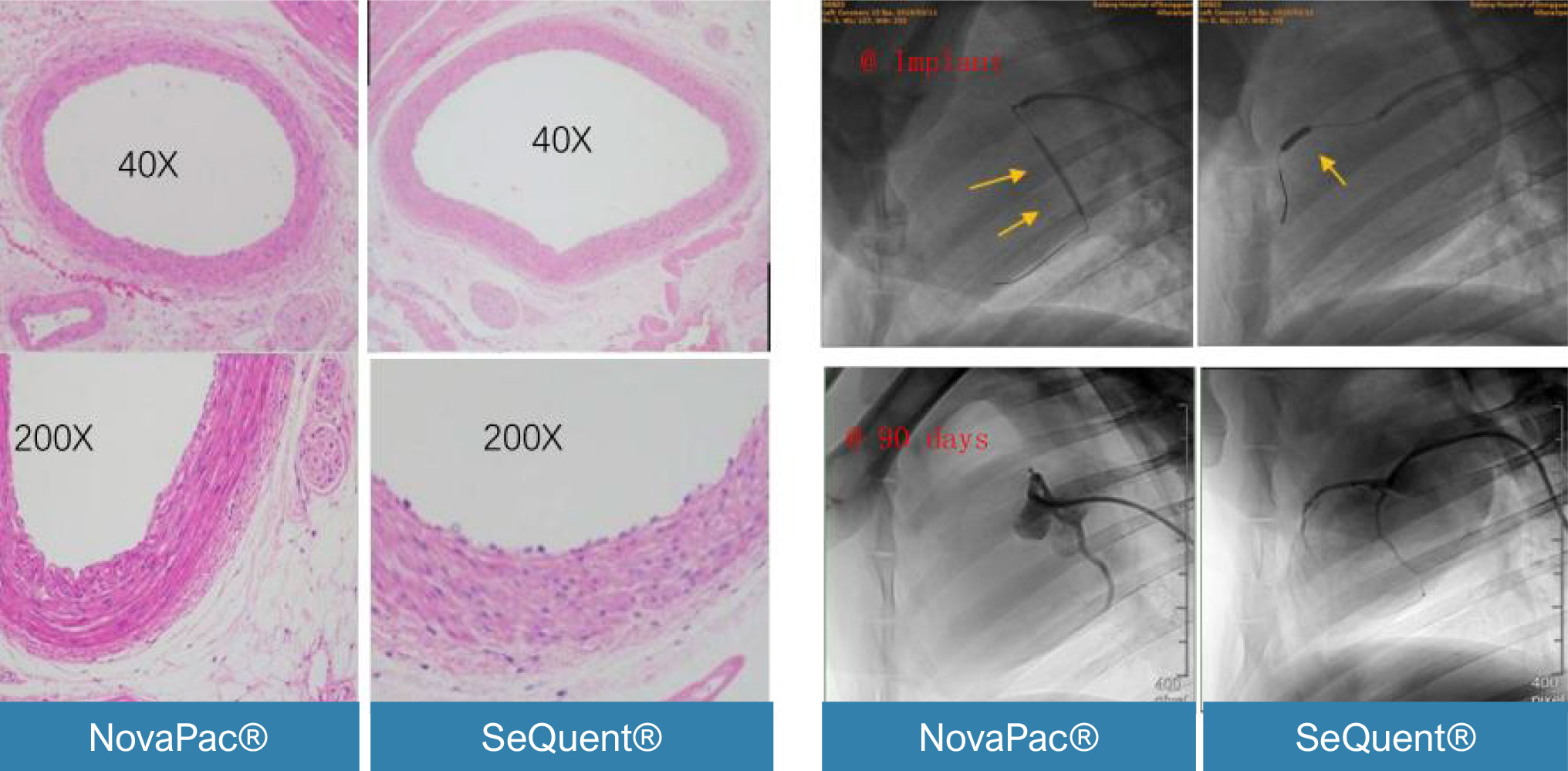
QCA and Histomorphometric comparison between SeQuent & NovaPac in porcine coronary arteries 90days post expansion:
There are no significant different.
(LA:Lumen Area; EA:internal Elastic Area; MA: Media Area; NIA: Neointima Area; RFD: vessel reference diameter; MLD: minimal lumen diameter; Lumen loss(%ds)=LLL/MLD post x 100%.(inside Data).
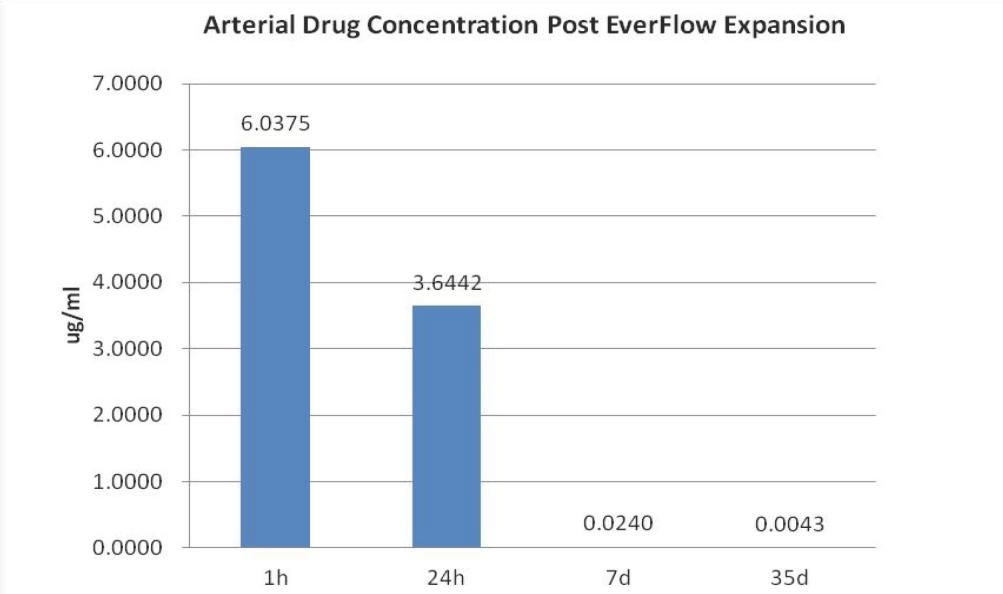
Coronary Vessel Paclitaxel Residual Dosage
Sufficient and remain up to 35 days post balloon expansion
(Paclitaxel safe & efficacy concentration: 0.0085-0.85ug/mL Avel et al: Circulation.1997; 96:636-645; Solot et al; J.Clin.Invest.1995;95:1869-1876)
NovaPac PTX Pharmacokinetics Study(PK) | ||||||
Tissues | Time Points(ug/g) | |||||
0 | 1h | 24h | 7 days | 35 days | ||
Blood | 2.62* | 0.0084±0.0043 | 0 | 0 | 0 | |
Vessels(Balloon Injured) | / | 6.04±1.78 | 3.64±2.21 | 0.024±.0082 | 0.0043±0.0018 | |
Core Life Organs | Myocardium | 0.21 | 0.006 | 0 | 0 | |
Liver | 0 | 0 | 0 | 0 | ||
Spleen | 0 | 0 | 0 | 0 | ||
Lung | 0 | 0 | 0.0091 | 0 | ||
Kidney | 0 | 0 | 0 | 0 | ||
Brain | 0 | 0 | 0 | 0 | ||
NovaPac PK Data
Paclitaxel (PTX) was detected only within one week in the core organs post balloon expansion, and within safe range (Inside data)
Order lnformation
NovaPac Paclitaxel-Coated PTCA Balloon Catheter(sizes) | ||||||||
Balloon | Balloon Length(mm) | |||||||
10 | 14 | 18 | 22 | 26 | 30 | 34 | 38 | |
2.00 | PCB20010 | PCB20014 | PCB20018 | PCB20022 | PCB20026 | PCB20030 | PCB20034 | PCB20038 |
2.25 | PCB22510 | PCB22514 | PCB22518 | PCB22522 | PCB22526 | PCB22530 | PCB22534 | PCB22538 |
2.50 | PCB25010 | PCB25014 | PCB25018 | PCB25022 | PCB25026 | PCB25030 | PCB25034 | PCB25038 |
2.75 | PCB27510 | PCB27514 | PCB27518 | PCB27522 | PCB27526 | PCB27530 | PCB27534 | PCB27538 |
3.00 | PCB30010 | PCB30014 | PCB30018 | PCB30022 | PCB30026 | PCB30030 | PCB30034 | PCB30038 |
3.25 | PCB32510 | PCB32514 | PCB32518 | PCB32522 | PCB32526 | PCB32530 | PCB32534 | PCB32538 |
3.50 | PCB35010 | PCB35014 | PCB35018 | PCB35022 | PCB35026 | PCB35030 | PCB35034 | PCB35038 |
3.75 | PCB37510 | PCB37514 | PCB37518 | PCB37522 | PCB37526 | PCB37530 | PCB37534 | PCB37538 |
4.00 | PCB40010 | PCB40014 | PCB40018 | PCB40022 | PCB40026 | PCB40030 | PCB40034 | PCB40038 |
CAUTION:
The law restricts these devices to sale by or on the order of a physician.Indications,contraindications, warnings and instructions for use can be found in the product labeling supplied with each device.
NovaPac Indications
A. De novo lesions (primary use in the case of stenosis or occlusion) including small vessels (SVD)
B. Residual stenosis after balloon or stent PTCA
C. Pre and post dilatation during coronary stent implantation
D. Acute or impending vascular occlusion
We pioneer polymer-free drug delivery innovation, with our NovaPac catheter avoiding polymer-related long-term vascular complications—a key advantage over traditional drug-coated balloons with polymer carriers. Our product is backed by evidence-driven safety and efficacy, with porcine coronary artery studies confirming non-inferior performance versus leading competitors, along with sustained drug retention and minimal systemic exposure. The physician-centric design, featuring a low-profile catheter and compatibility with standard 0.014-inch guide wires, streamlines procedural workflows and reduces intervention time.
The catheter delivers paclitaxel that remains therapeutically active in the target vessel for up to 30 days post-balloon expansion.
Yes, it is available in diameters as small as 2.00mm, making it ideal for treating small coronary artery lesions.
The catheter features a semi-compliant balloon with a 100% paclitaxel coating and a lubricious shaft for enhanced deliverability, with no polymer carriers.
