| Availability: | |
|---|---|
| Quantity: | |











NovaPac Paclitaxel-Coated PTCA Balloon Catheter is a cutting-edge coronary intervention device integrating the Lumineer drug delivery-specific balloon catheter and polymer-free paclitaxel drug coating. Engineered for precise drug and stent delivery, it features semi-compliant properties, excellent crossability, and accurate balloon expansion. The device adopts special coating technology to achieve 100% uniform surface coating, precision drug loading, and controlled drug release, ensuring minimal drug loss during delivery and sustained release during balloon expansion. Clinical data proves its long-last vessel drug retention and efficacy, with effective drug dose remaining in vessels for up to 30 days post-expansion, making it a reliable choice for coronary artery disease treatment.
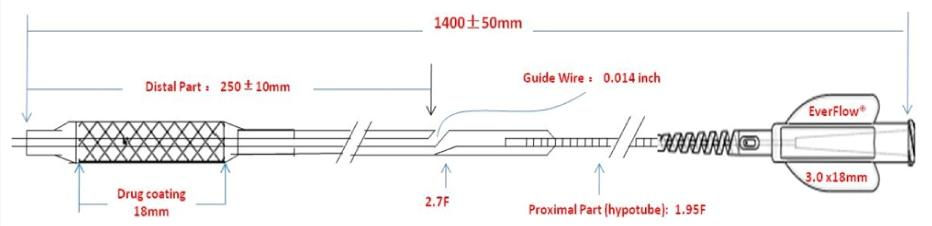
Distal part length: 250±10mm
Guide wire specification: 0.014 inch
Proximal hypotube size: 1.95F
The 100% uniform balloon surface coating ensures high drug adhesion and transfer efficiency to vessels. The controlled release profile minimizes drug loss during the delivery process, maximizing the effective drug concentration acting on target lesions.
Pharmacokinetic studies show that paclitaxel residual dosage in coronary vessels remains sufficient for up to 35 days post balloon expansion, well within the safe and effective concentration range (0.0085-0.85ug/mL). This long retention period enhances the device’s therapeutic effect on vascular lesions.
Histomorphometric analysis at 90 days post-expansion shows no significant difference compared with the benchmark product. Paclitaxel is only detected in core organs within one week post-operation and stays in the safe range, eliminating potential systemic side effects.
It is primarily indicated for primary stenosis or occlusion lesions, including small vessel disease (SVD), effectively dilating narrow vessels and delivering drugs to prevent restenosis.
The device can handle residual stenosis after conventional balloon or stent PTCA procedures, further optimizing the luminal diameter of blood vessels.
It serves as a critical auxiliary tool for coronary stent implantation, dilating vessels before stent placement and optimizing stent apposition after implantation.
It provides emergency treatment for acute or impending vascular occlusion, quickly restoring blood flow in blocked vessels.
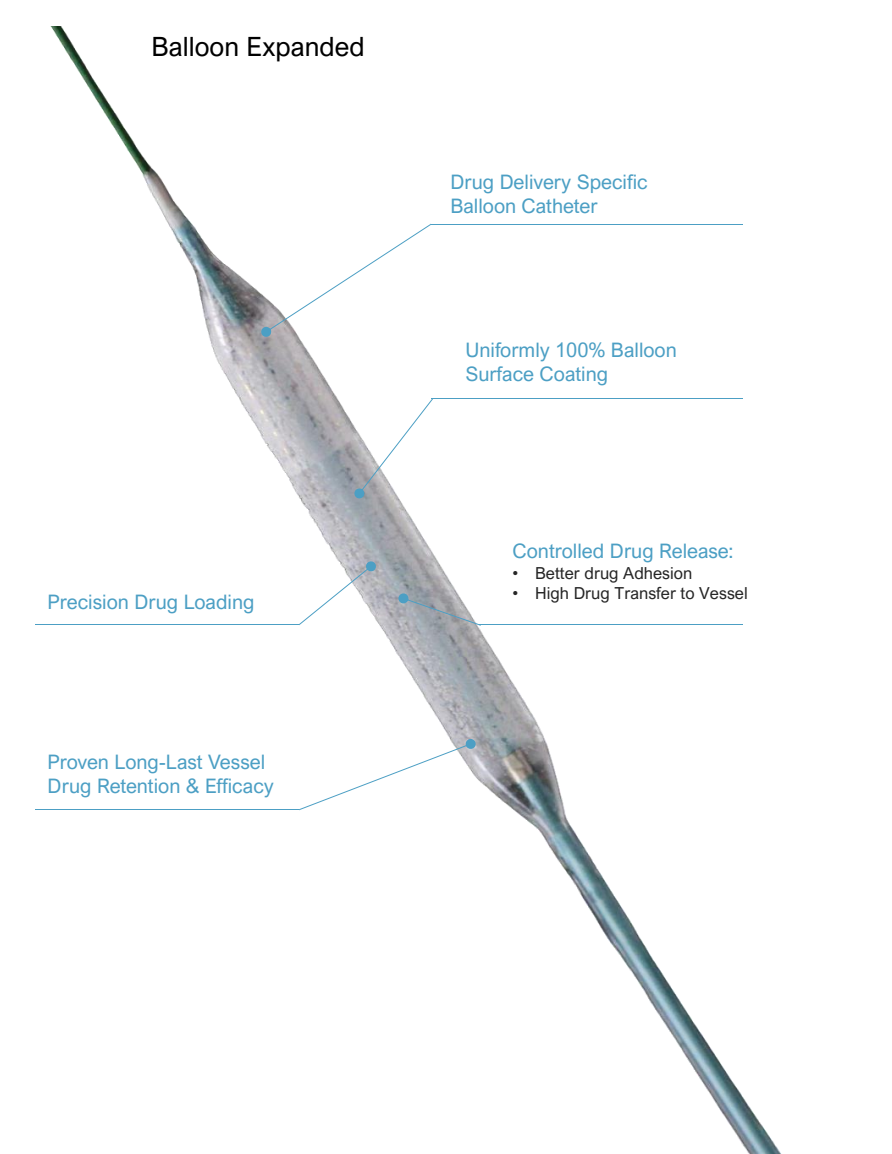
Evidence-Driven Device Safety and Efficacy
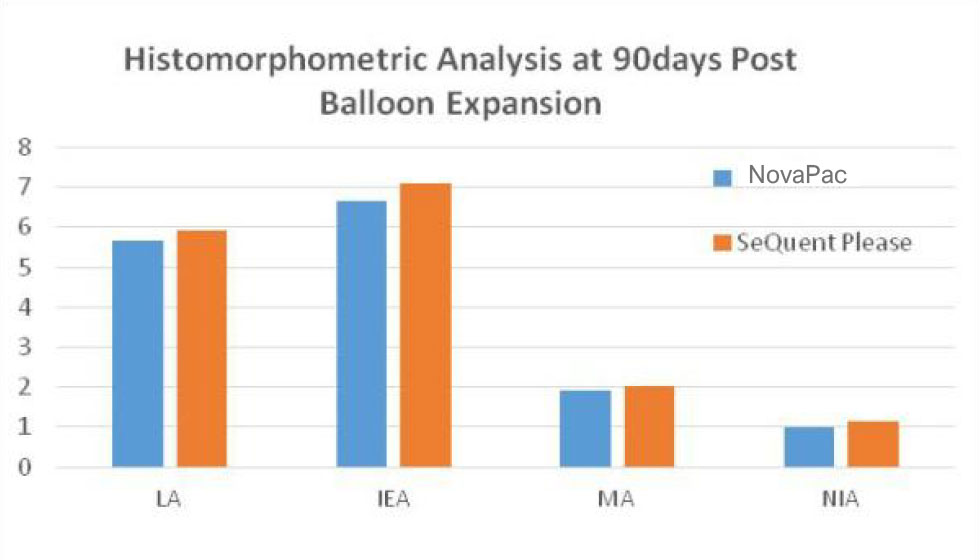
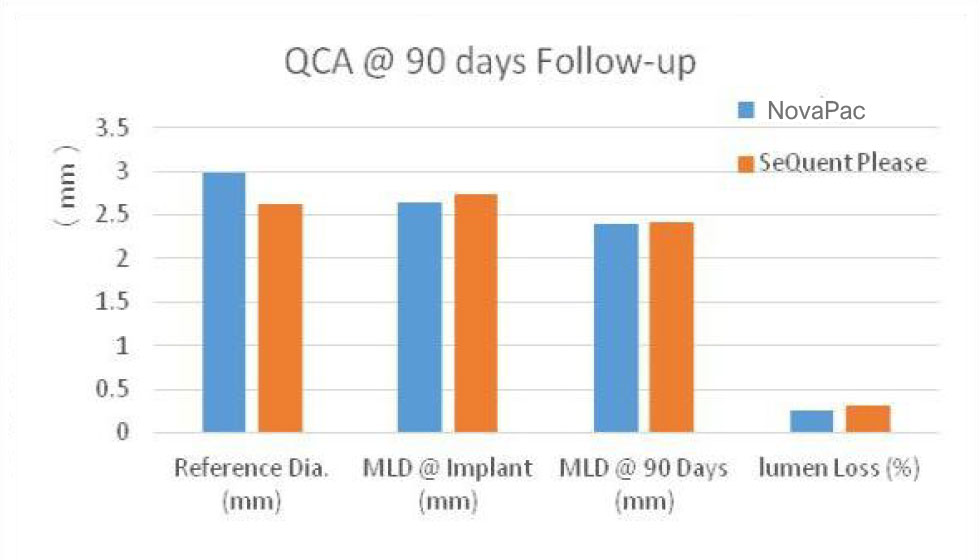
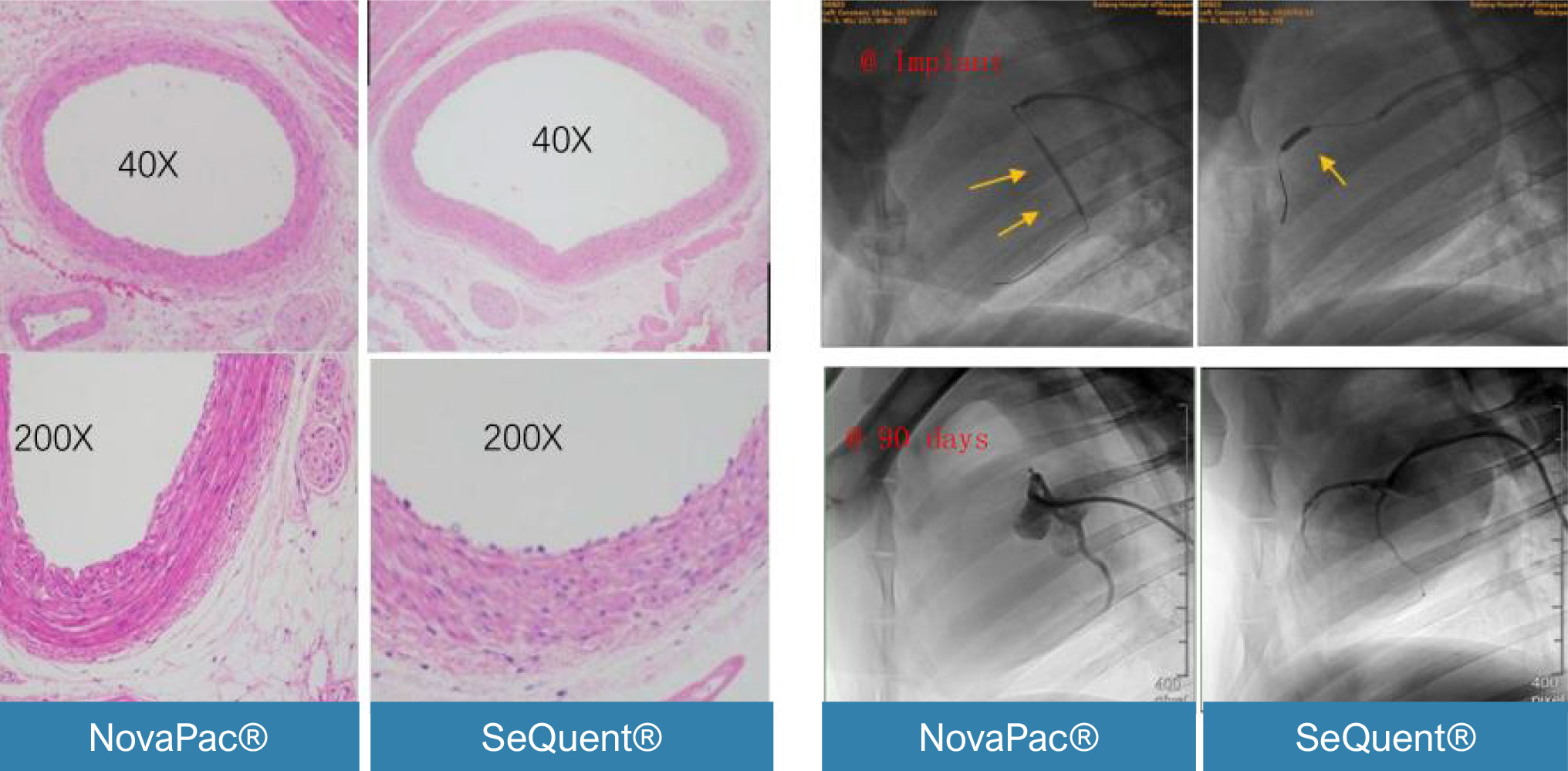
QCA and Histomorphometric comparison between SeQuent & NovaPac in porcine coronary arteries 90days post expansion:
There are no significant different.
(LA:Lumen Area; EA:internal Elastic Area; MA: Media Area; NIA: Neointima Area; RFD: vessel reference diameter; MLD: minimal lumen diameter; Lumen loss(%ds)=LLL/MLD post x 100%.(inside Data).
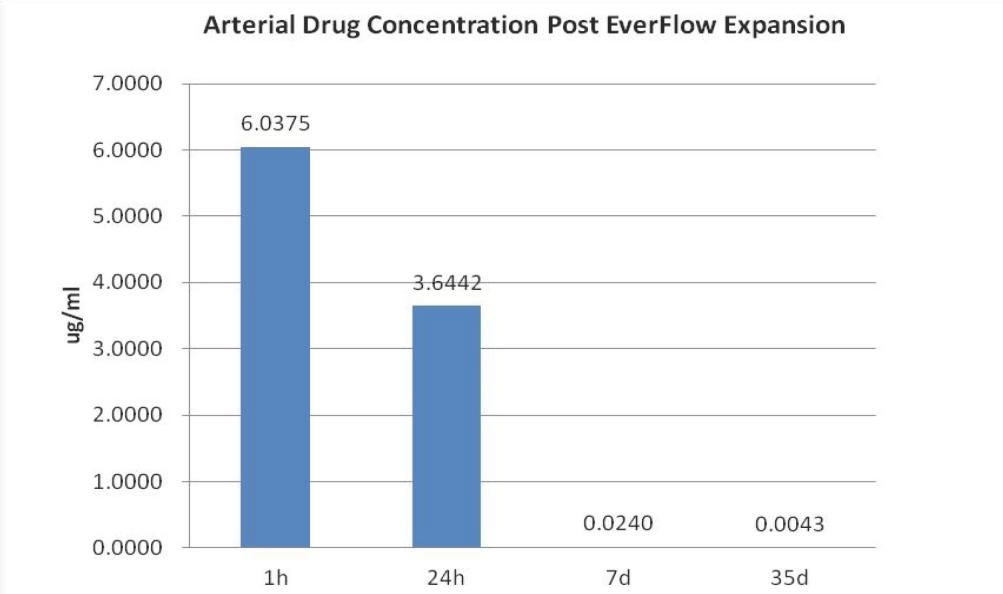
Coronary Vessel Paclitaxel Residual Dosage
Sufficient and remain up to 35 days post balloon expansion
(Paclitaxel safe & efficacy concentration: 0.0085-0.85ug/mL Avel et al: Circulation.1997; 96:636-645; Solot et al; J.Clin.Invest.1995;95:1869-1876)
NovaPac PTX Pharmacokinetics Study(PK) | ||||||
Tissues | Time Points(ug/g) | |||||
0 | 1h | 24h | 7 days | 35 days | ||
Blood | 2.62* | 0.0084±0.0043 | 0 | 0 | 0 | |
Vessels(Balloon Injured) | / | 6.04±1.78 | 3.64±2.21 | 0.024±.0082 | 0.0043±0.0018 | |
Core Life Organs | Myocardium | 0.21 | 0.006 | 0 | 0 | |
Liver | 0 | 0 | 0 | 0 | ||
Spleen | 0 | 0 | 0 | 0 | ||
Lung | 0 | 0 | 0.0091 | 0 | ||
Kidney | 0 | 0 | 0 | 0 | ||
Brain | 0 | 0 | 0 | 0 | ||
NovaPac PK Data
Paclitaxel (PTX) was detected only within one week in the core organs post balloon expansion, and within safe range (Inside data)
Order lnformation
NovaPac Paclitaxel-Coated PTCA Balloon Catheter(sizes) | ||||||||
Balloon | Balloon Length(mm) | |||||||
10 | 14 | 18 | 22 | 26 | 30 | 34 | 38 | |
2.00 | PCB20010 | PCB20014 | PCB20018 | PCB20022 | PCB20026 | PCB20030 | PCB20034 | PCB20038 |
2.25 | PCB22510 | PCB22514 | PCB22518 | PCB22522 | PCB22526 | PCB22530 | PCB22534 | PCB22538 |
2.50 | PCB25010 | PCB25014 | PCB25018 | PCB25022 | PCB25026 | PCB25030 | PCB25034 | PCB25038 |
2.75 | PCB27510 | PCB27514 | PCB27518 | PCB27522 | PCB27526 | PCB27530 | PCB27534 | PCB27538 |
3.00 | PCB30010 | PCB30014 | PCB30018 | PCB30022 | PCB30026 | PCB30030 | PCB30034 | PCB30038 |
3.25 | PCB32510 | PCB32514 | PCB32518 | PCB32522 | PCB32526 | PCB32530 | PCB32534 | PCB32538 |
3.50 | PCB35010 | PCB35014 | PCB35018 | PCB35022 | PCB35026 | PCB35030 | PCB35034 | PCB35038 |
3.75 | PCB37510 | PCB37514 | PCB37518 | PCB37522 | PCB37526 | PCB37530 | PCB37534 | PCB37538 |
4.00 | PCB40010 | PCB40014 | PCB40018 | PCB40022 | PCB40026 | PCB40030 | PCB40034 | PCB40038 |
CAUTION:
The law restricts these devices to sale by or on the order of a physician.Indications,contraindications, warnings and instructions for use can be found in the product labeling supplied with each device.
NovaPac Indications
A. De novo lesions (primary use in the case of stenosis or occlusion) including small vessels (SVD)
B. Residual stenosis after balloon or stent PTCA
C. Pre and post dilatation during coronary stent implantation
D. Acute or impending vascular occlusion
QCA and histomorphometric comparison in porcine coronary arteries prove that NovaPac has equivalent performance to mainstream products, with reliable clinical data support.
The polymer-free paclitaxel coating avoids the potential inflammatory response caused by polymer materials, reducing the risk of post-operative complications.
With balloon diameters ranging from 2.00mm to 4.00mm and 8 length specifications, NovaPac can meet the treatment needs of different lesion sizes and positions.
The core advantage lies in 100% uniform surface coating and polymer-free design. It achieves high drug adhesion, controlled release, and long-term retention, while reducing polymer-related side effects.
NovaPac is suitable for most common coronary lesion scenarios, including de novo lesions, residual stenosis, and stent-related dilatation. Specific indications should comply with the product labeling and physician’s judgment.
